Elements
Elements are the furthest something can be broken down into. They're the simplest form of matter. Building blocks for all living things
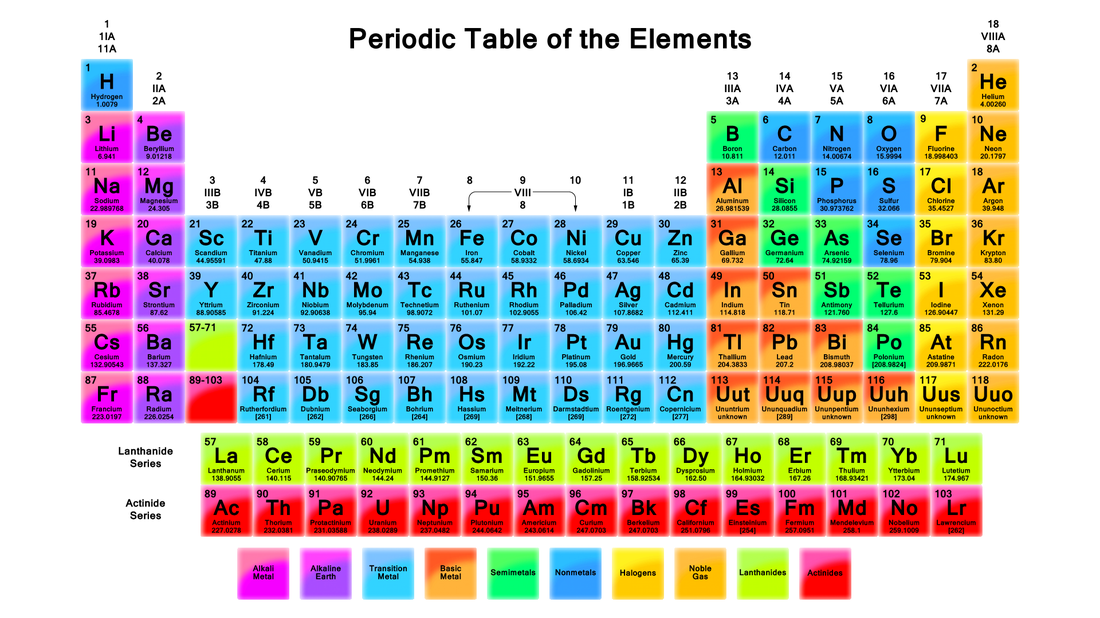
There are 117 different elements. Their are 92 found in nature, these are found on the table above. They're 25 that are created by Humans, and these are not on the Periodic table. The Majority of your body is made of Oxygen, Carbon, Hydrogen and Nitrogen.
Atomic Structure: Made of Protons (Positive Charge) Electrons (Negative Charge) and Neutrons (Neutral Charge)
Number of Protons is equal to number of electrons, and each atom wants a total of 8 protons and 8 electrons
Covalent Ionic same but different.
Atomic Structure: Made of Protons (Positive Charge) Electrons (Negative Charge) and Neutrons (Neutral Charge)
Number of Protons is equal to number of electrons, and each atom wants a total of 8 protons and 8 electrons
Covalent Ionic same but different.
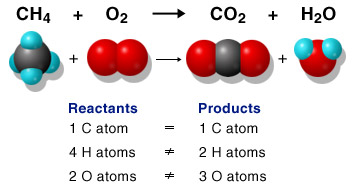
A chemical reaction is when two different elements are bonded or split into parts.
A Molecule is any group of more than one atom of an element, or an atom of more than one element
An Ionic bond is when two elements are bonded by one element taking protons from another element so that both elements have a full ring or a full orbit
A Covalent Bond is where two elements use the others Protons to get a full orbit, so they are the protons. Covalent Bonds are also typically between the same element.
A Molecule is any group of more than one atom of an element, or an atom of more than one element
An Ionic bond is when two elements are bonded by one element taking protons from another element so that both elements have a full ring or a full orbit
A Covalent Bond is where two elements use the others Protons to get a full orbit, so they are the protons. Covalent Bonds are also typically between the same element.